Introduction: A Historic Moment for Mental Health
Imagine treating depression without pills. No weight gain. No sexual dysfunction. No emotional blunting. Just a gentle current to your brain while you sit at home.
This is not science fiction. This is December 8, 2025—the day the FDA approved the first at-home brain stimulation device for major depression . electric medicine, depression, 2026
For the first time in history, millions of Americans will soon have access to a prescription device that treats depression through electricity rather than medication. It’s called the Flow FL-100, and it arrives in the US market in spring 2026 .
This article explores this revolutionary technology, how it works, what science says about its effectiveness, and whether it might replace antidepressants for millions of people.
—
The Depression Crisis That Demands New Solutions
Alarming Statistics
Depression rates have reached unprecedented levels:
· 20+ million US adults currently live with depression
· 60% increase over the past decade
· One-third of patients do not respond adequately to antidepressant medications
· Many others discontinue medication due to side effects:
· Sexual dysfunction
· Weight gain
· Emotional blunting
· Sleep disturbances
Depression doesn’t exist in isolation—it often connects with other mental health challenges. If you’re struggling with low mood, you might also be experiencing stress and anxiety. Our comprehensive guide on stress, anxiety, and depression explains the differences between these conditions and offers evidence-based coping strategies that complement professional treatment.
The Treatment Gap
Dr. Samuel Wilkinson, associate professor of psychiatry at Yale Medicine, explains: “While medications and talk therapy are effective for many people, symptom relief can be elusive” . Approximately one-third of patients either don’t respond to antidepressants or can’t tolerate their side effects .
This treatment gap has created urgent demand for alternatives—and now, technology is delivering one.
—

—
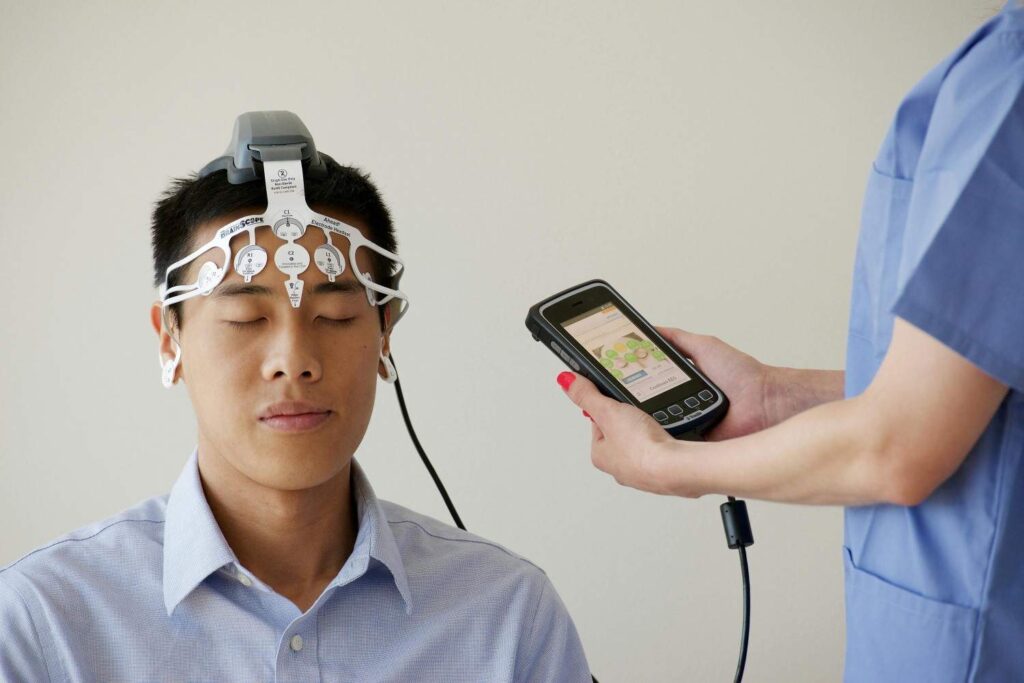
What Is the Flow FL-100?
The Flow FL-100 is a wireless headset that delivers transcranial Direct Current Stimulation (tDCS) —a gentle electrical current to the brain’s prefrontal cortex .
How It Works
The prefrontal cortex is the brain region responsible for:
· Mood regulation
· Stress response
· Emotional control
· Decision-making
In people with depression, this area is often underactive . The FL-100 delivers a low-intensity electrical current (0.5 to 2 milliamperes) through two electrodes placed on the scalp .
How weak is this current? For comparison:
· tDCS (Flow FL-100): 0.5–2 mA (gentle tingling)
· Electroconvulsive therapy (ECT): 800 mA (seizure-inducing)
“The FL-100 works by delivering a gentle, comfortable electrical signal to the forehead to restore healthy brain activity and reduce symptoms,” explains Dr. Kultar Garcha, Flow’s chief medical officer .
What the Device Includes
· Wireless headset with two electrodes
· Companion iOS/Android app
· Remote monitoring by healthcare providers
· Prescription required from licensed clinician
—
The Science Behind the Device
25+ Years of Research
tDCS technology isn’t new. It has accumulated more than 25 years of clinical investigation across depression, stroke rehabilitation, pain conditions, and other neurologic indications, with over 9,000 publications .
The Pivotal Nature Medicine Trial
The FDA approval was based on a landmark study published in Nature Medicine—one of the world’s most prestigious scientific journals .
Study design:
· 174 adults with moderate to severe major depression
· Randomized, double-blind, sham-controlled (placebo) trial
· 10-week treatment course
· Remote supervision at home
Treatment protocol:
Weeks Sessions
First 3 weeks 5 sessions per week (30 minutes each)
Next 7 weeks 3 sessions per week (30 minutes each)
Results That Changed Mental Health Care
After 10 weeks of use, the results were remarkable :
Outcome Active tDCS Group Placebo Group
Treatment response 45% 22%
Remission (symptoms gone) 58% Significantly lower
Improvement speed 3-4 weeks Slower
Researchers measured improvements in:
· Mood
· Energy levels
· Sleep quality
· Concentration
· Anxiety
Statistical significance: Active tDCS produced a mean HDRS improvement of 9.41 points versus 7.14 points in the placebo group (P = 0.012) .
The remarkable results of brain stimulation therapy highlight how directly targeting brain activity can improve mental health. This aligns with what we know about emotional fitness—the ability to understand and regulate your emotional states. Our guide to emotional fitness and mental strength provides complementary practices that can enhance any depression treatment.
Real-World Results
More than 55,000 people in Europe, the UK, Switzerland, and Hong Kong have already used the system, including within several NHS trusts .
“Among our real world users, 77% see improvements in as little as three weeks,” reports Dr. Garcha .
—
—
A Second Option: Neurolief’s ProlivRx
The Flow FL-100 isn’t the only device entering the market. In January 2026, the FDA approved ProlivRx by Neurolief—another at-home neuromodulation therapy .
How ProlivRx Differs
ProlivRx uses a different technology called eCOT-AS (external Combined Occipital and Trigeminal Afferent Stimulation) . It delivers gentle electrical pulses to:
· Occipital nerve pathways (back of head)
· Trigeminal nerve pathways (face)
These pathways conduct stimulation to the brainstem and higher brain regions involved in depression .
Clinical Evidence
The MOOD clinical trial (124 participants) showed :
· 21.3% remission rate with active treatment vs 6.0% with placebo
· Mean HDRS score reduction of 8.62 points vs 6.01 points
· At 16 weeks, remission rates increased to 31.7%
Who It’s For
ProlivRx is specifically indicated for adults with major depression who did not achieve adequate improvement with at least one antidepressant .
—
Side Effects and Safety
Flow FL-100 Side Effects
Reported side effects are generally mild and transient :
Side Effect Frequency
Tingling at electrode sites Common
Brief skin irritation Common
Skin redness after prolonged use Common
Transient headache Reported
Stinging or itching Reported
Important safety note: Skin burns have occurred only when patients reused electrodes or allowed electrode pads to dry out—which device instructions warn against .
The absence of systemic side effects with brain stimulation devices stands in stark contrast to many medications. But mental health isn’t just about treating depression—it’s about overall well-being. For a broader perspective on building mental resilience, explore our guide on the vagus nerve and how activating your body’s relaxation response can reduce stress and improve emotional balance.
ProlivRx Safety
ProlivRx was well-tolerated with mostly mild, transient adverse events. The most common was headache (5 participants). No systemic adverse effects were reported .
No Systemic Side Effects
Unlike antidepressants, these devices avoid :
· ❌ Weight gain
· ❌ Sexual dysfunction
· ❌ Emotional blunting
· ❌ Gastrointestinal issues
—
Who Is a Good Candidate?
Flow FL-100 Candidates
The FL-100 is indicated for adults (18+) with :
· Moderate to severe major depressive disorder
· Can be used as monotherapy (alone) or adjunctive (with medication)
· Not considered “treatment refractory”
Dr. Garcha notes: “It fits well when a patient has experienced a recent change in their condition or life, suggesting their brain is ‘primed for plasticity,’ meaning it is adaptable and ready to relearn healthy patterns” .
ProlivRx Candidates
ProlivRx is for adults with MDD who failed to achieve satisfactory improvement from at least one previous antidepressant .
Who Should NOT Use These Devices
· Individuals with treatment-resistant depression (specific criteria)
· People with implanted medical devices (consult physician)
· Those with certain neurological conditions
Always consult a healthcare provider before starting any new treatment.
—

—
Availability and Cost
Flow FL-100 Availability
· US Launch: Q2 2026 (spring/summer)
· Availability: By prescription only
· App Stores: iOS and Android apps will be available for download
Cost
Flow expects the US retail price to be between $500 and $800 . Pricing for electrode pads or potential subscription components hasn’t been released.
Insurance Coverage
Flow is “actively engaging in discussions with a number of U.S. insurance payers” . However, “it’s unlikely that broad coverage will be in place at the Q2 launch” .
Erin Lee, Flow’s CEO, states: “We’re on a mission to make effective, affordable non-drug treatment available to the millions of Americans suffering from depression” .
—
Will This Replace Antidepressants?
Expert Opinions
Dr. Samuel Wilkinson (Yale Medicine): “It’s another arrow in the quiver — an option that appears helpful for a meaningful subset of patients” .
Dr. Linda Carpenter (Brown University): “Even though there is a tremendous need for new treatment approaches when antidepressant medications aren’t working, access to interventional psychiatry services like Transcranial Magnetic Stimulation (TMS) remains limited. Now we can offer our patients ProlivRx therapy with continued medical supervision and the convenience of home use” .
A Paradigm Shift
Erin Lee, Flow’s CEO, calls this “a watershed moment for the treatment of depression: the first step in moving from pharmaceutical treatments to tech-based therapies with minimal side effects” .
Realistic Perspective
These devices will likely:
· ✅ Supplement rather than completely replace medication
· ✅ Offer hope to those who don’t respond to drugs
· ✅ Provide options for those who can’t tolerate side effects
· ✅ Expand access to millions who lack clinic-based care
—
The Future of Brain Stimulation
Flow plans to evaluate its platform for additional conditions :
· Traumatic brain injury
· Addiction
· Sleep disorders
Neurolief is also exploring broader applications .
Dr. Marom Bikson, writing in the journal Brain Stimulation, states: “This is a major regulatory milestone in non-pharmacological treatment of depression, and paves the way for additional uses and new technologies of home-based neuromodulation” .
—
Common Questions About Brain Stimulation Devices
Is this the same as electroshock therapy?
Absolutely not. ECT delivers about 800 milliamperes and intentionally causes seizures. tDCS delivers 0.5–2 milliamperes—a fraction of the strength—and you remain fully awake and aware .
Does it hurt?
Most users report a mild tingling or itching sensation at the electrode sites during the first few minutes. This typically fades as treatment continues .
How long until I see results?
Clinical trial data shows improvements as early as 3-4 weeks of consistent use .
Can I use it with my current medication?
Yes. The FL-100 is approved as both monotherapy (alone) and adjunctive treatment (with medication). Clinical trials included participants receiving concurrent pharmacotherapy .
Do I need a prescription?
Yes. Both devices require a prescription from a licensed healthcare provider .
Will insurance cover it?
Not immediately at launch, but manufacturers are actively negotiating with insurance providers .
—
Conclusion: A New Era for Mental Health
December 8, 2025, will be remembered as the day mental health treatment changed forever.
For the first time, millions of Americans will have access to a scientifically proven, FDA-approved, non-drug treatment for depression that they can use in their own homes. No clinic visits. No systemic side effects. Just gentle brain stimulation while reading a book or watching television.
This doesn’t mean antidepressants will disappear. Medications will remain essential for many people. But for the one-third who don’t respond to drugs, for those who can’t tolerate side effects, and for those seeking alternatives, electric medicine has finally arrived.
As Erin Lee, Flow’s CEO, put it: “We’re on a mission to make effective, affordable non-drug treatment available to the millions of Americans suffering from depression” .
That mission has just begun.
The arrival of electric medicine represents a paradigm shift in how we approach mental health. But technology alone isn’t enough—sustainable wellness requires a holistic approach. Our complete guide to long-term healthy lifestyle choices brings together nutrition, physical activity, stress management, and emotional resilience to help you build a foundation for lasting mental and physical health.
—
Disclaimer
Important Medical Disclaimer
This content is for general informational purposes only and does not constitute medical, health, or professional advice. The information provided is based on research available as of early 2026, including FDA announcements and published clinical trials.
Always consult a qualified healthcare professional before starting any new treatment for depression or other medical conditions. Never stop or change antidepressant medications without consulting your healthcare provider.
Individual responses to brain stimulation therapy vary significantly. Results reported in clinical trials may not predict individual outcomes. Side effects, while generally mild, should be discussed with a healthcare provider.
Medical devices mentioned (Flow FL-100, ProlivRx) require a prescription and are not available over the counter. Insurance coverage and pricing information is subject to change.
